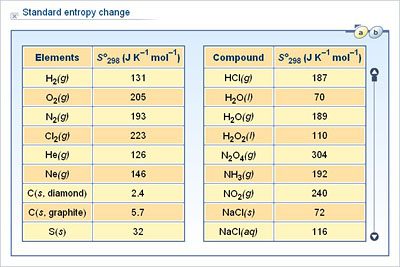
The entropy of a pure crystalline substance at absolute zero is defined to be equal to zero. As the temperature of the substance increases, its entropy increases because of an increase in molecular motion. Note: The standard entropy change is equal to the sum of all the standard entropies of the products minus the sum of all the standard entropies of the reactants. In chemistry, the standard molar entropy is the entropy content of one mole of pure substance at a standard state of pressure and any temperature of. The value for S rxn is negative, as expected for this phase transition (condensation). We have an Answer from Expert Buy This Answer 5 Place Order. 2Al(s) + 3FeO(s) -> Al 2 O 3 (s) + 3Fe(s) We have an Answer from Expert View Expert Answer.  
Round your answer to 4 significant digits. It has found far-ranging applications in chemistry and physics, biological systems and their relation to life, cosmology, economics, sociology, weather science and climate change, and information system and the transmission of information in telecommunication. The standard entropy changes for the reaction, S rxn is calculated using the standard molar entropies and stoichiometric coefficients. Calculate the standard entropy change for the following reaction at 25.0 C. The term and the concept are used in diverse fields, from classical thermodynamics, where it was first recognized, to the microscopic description of nature in statistical physics, and to the principles of information theory. To calculate entropy first we need to know what it actually stands for, entropy is a scientific concept, as well as a measurable physical property that is most commonly associated with a state of disorder, randomness, or uncertainty. According to the second law of thermodynamics, a spontaneous reaction always results in an increase in total entropy of the system and its surroundings. Determine if the entropy change will be positive or negative for the following reactions: A) (NH4)2Cr2O7 (s) Cr2O3 (s) + 4 H2O (l) + CO2 (g) B) 2 H2 (g) + O2 (g) 2 H2O (g) C) PCl5 PCl3 + Cl2 (g) Solution The entropy of a reaction refers to the positional probabilities for each reactant. Under identical conditions, it is greater for a heavier gas. The standard entropy changes apparently increased with the heats of formation for the alloy with different composition. Same old same old - this is just products minus reactants. Hint: The standard entropy of reaction helps us determine whether the reaction will take place or not because molar entropy is not the same for all gases. 15.2 Calculate the standard entropy change for a reaction using standard entropy values.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
